
A compound is said to be soluble if it readily dissolves in water and does not precipitate if left undisturbed for an extended period of time. New Concepts: An aqueous solution is a solution with water as the solvent. Write net ionic equations given the reactant salts. Correctly predict the products of a double replacement reaction. Write simple net ionic equations for double replacement reactions. Solubility Rules and Net Ionic Equations Objective: Develop and utilize solubility rules for common ions in water. Stephen Prilliman, Harding Charter Preparatory High School E-mail: Author grants the right to copy and edit for educational purposes Model I: Rules of Solubility in Aqueous Solutions POGIL Workshop Activity © Dr. A compound is said to be soluble if it readily dissolves in water and does not fall out of solution ( precipitate ) if left for an extended period of time. An aqueous solution is a solution where the solvent is water. Whatever is dissolved in the liquid is called the solute. The liquid present in greatest amount is called the solvent.

Definitions A solution is a homogeneous mixture of a solid, liquid or gas in a liquid.
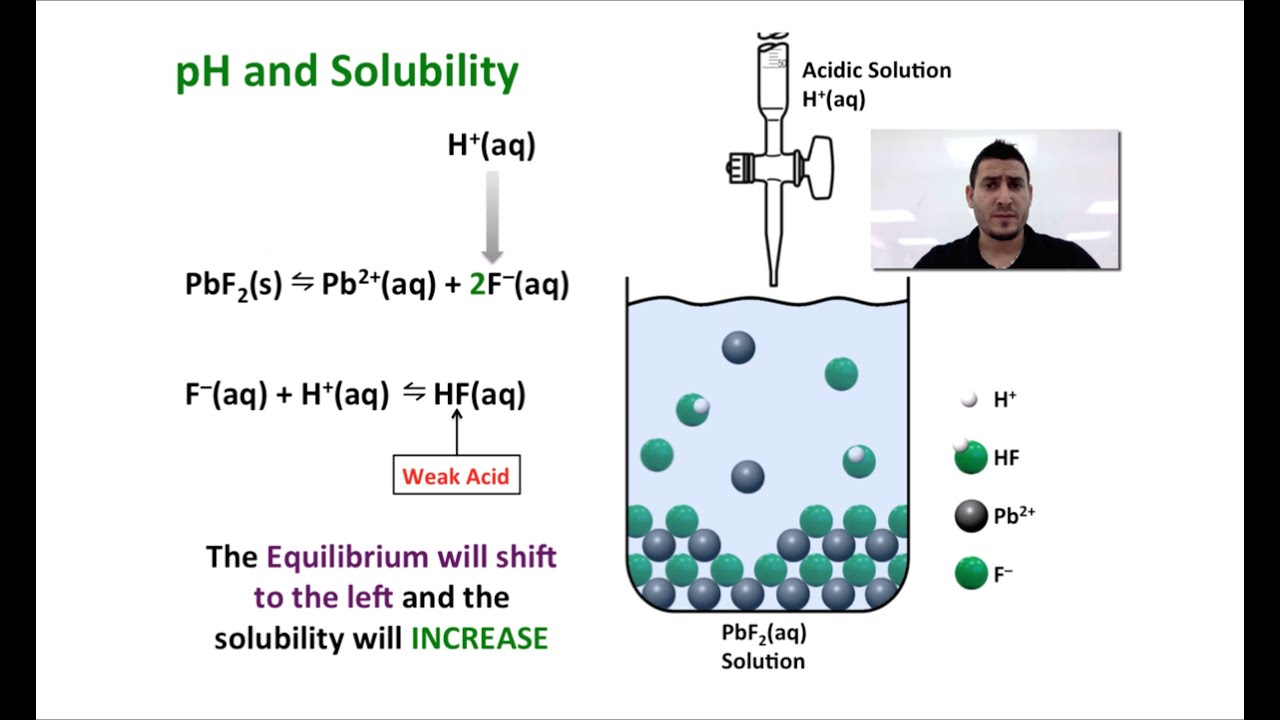
Before you begin you should understand: ➢ Naming of binary compounds ➢ Writing chemical formulae ➢ Predicting products for double displacement reactions Before you leave today you should be able to: ➢ Write simple net ionic reactions given any two reactants. We resolve this conundrum by using a notation called net ionic equations that we will learn about in this activity. When aqueous solutions of compounds are mixed some reactions are observed to occur while other do not. Sometimes our “paper and pencil” chemistry disagrees with what we see in the lab. Reactions in Water (Solubility Rules) Complete and Net Ionic Equations A POGIL activity WHY? Writing reaction equations is a way of predicting what reactions will take place between two substances. These are general rules, based on observation.Solubility can be predicted from rules (pg.399).Soluble and insoluble are general terms to.Precipitation refers to the formation of a solid.Writeīalanced molecular, ionic & net ionic equations To get the NET ionic equation we cancel out.

- We can write this as an ionic equation (allĬompounds that are (aq) are written as ions):īoth sides of a reaction should have the same.
- There are two conditions for molecular, ionic,īoth sides of an equation should have the.
- Has a subscript of 2 in the original formulaĮquations can be divided into 3 types (pg.































 RSS Feed
RSS Feed
